Đề thi thử THPT Quốc gia năm 2017 môn Hóa học Sở GD&ĐT tỉnh Đồng Tháp
- Thứ ba - 23/05/2017 10:00
- In ra
- Đóng cửa sổ này
Đề thi thử THPT Quốc gia năm 2017 môn Hóa học Sở GD&ĐT tỉnh Đồng Tháp, có đáp án kèm theo. Mời các bạn cùng tham khảo.
| SỞ GIÁO DỤC VÀ ĐÀO TẠO TỈNH ĐỒNG THÁP | THI DIỄN TẬP THPT QUỐC GIA NĂM HỌC 2016 - 2017 | |
| Mã đề 132 | ||
| ĐỀ THI MÔN: HÓA HỌC Ngày thi: 16 /05 /2017 Thời gian làm bài: 50 phút (Không kể thời gian phát đề) (Đề thi gồm có: 04 trang) | ||
Cho biết nguyên tử khối của các nguyên tố: H =1; C = 12; N = 14; O = 16; Na = 23; Mg = 24; Al = 27; S =32; Cl = 35,5; K = 39; Ca = 40; Cr = 52; Fe = 56; Cu = 64; Zn = 65; Ag = 108; Ba=137; Br = 80
Câu 1: Tiến hành các thí nghiệm sau:
(a). Cho lá Fe vào dung dịch gồm CuSO4 và H2SO4 (loãng)
(b). Đốt dây Fe trong bình đựng khí O2
(c). Cho lá Cu vào dung dịch hỗn hợp gồm Fe(NO3)3 và HNO3
(d). Cho lá Zn vào dung dịch HCl
Số thí nghiệm có xảy ra ăn mòn điện hoá là
A. 1 B. 2 C. 3 D. 4
Câu 2: Sắt bị ăn mòn điện hoá khi tiếp xúc với kim loại M để ngoài không khí ẩm. Vậy M là
A. Mg B. Zn C. Cu D. Al
Câu 3: Kim loại nào sau đây là kim loại kiềm thổ?
A. Na B. Al C. Ca D. K
Câu 4: Hòa tan hoàn toàn 4,36 gam hỗn hợp gồm K, Na, Ca và Ba trong nước dư thu được 1,232 lít H2 (đktc) và dung dịch X. Trung hòa X bằng dung dịch HCl vừa đủ rồi cô cạn dung dịch thu được m gam hỗn hợp muối khan. Giá trị m là
A. 8,265 B. 9,012 C. 8,964 D. 7,920
Câu 5: Phát biểu không đúng là:
A. Cr2O3 tan trong dung dịch NaOH loãng, đun nóng
B. CrO3 bốc cháy khi tiếp xúc S, C, P, C2H5OH
C. Nhỏ dung dịch H2SO4 vào dung dịch K2CrO4, màu dung dịch chuyển từ vàng sang da cam
D. Cho CrO3 vào nước luôn thu được hỗn hợp 2 axit
Câu 6: Phương trình hoá học nào sau đây sai?
A. Fe + Cl2→ FeCl2 B. Fe + H2SO4 loãng → FeSO4 + H2
C. Cr(OH)2 + 2HCl→ CrCl2 + 2H2O D. Zn + 2CrCl3→ ZnCl2 + 2CrCl2
Câu 7: Amin dùng để điều chế nilon -6,6 có tên là
A. phenylamin B. benzylamin
C. hexylamin D. hexametylenđiamin
Câu 8: Trung hòa 11,8 gam một amin đơn chức X cần 200 ml dung dịch HCl 1M. Công thức phân tử của X là
A. C3H9N B. C2H7N C. CH5N D. C3H7N
Câu 9: Công thức phân tử este no đơn chức mạch hở là
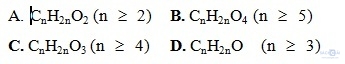
Câu 10: Cacbohiđrat X là chất dinh dưỡng và được dùng làm thuốc tăng lực. Cacbohiđrat X là
A. Xenlulozơ B. Fructozơ C. Glucozơ D. Saccarozơ
Câu 11: Kim loại có khối lượng riêng lớn nhất là
A. Li B. Mg C. Al D. Os
Câu 12: Chất chỉ có tính oxi hoá mạnh là
A. CrCl2 B. Cr2O3 C. CrO3 D. CrCl3
Câu 13: Kim loại chiếm khoảng 5% khối lượng vỏ Trái Đất, đứng hàng thứ hai sau nhôm là
A. Ca B. Cr C. Fe D. Mg
Câu 14: Thuỷ ngân dễ bay hơi và rất độc. Nếu chẳng may nhiệt kế thuỷ ngân bị vỡ thì dùng chất nào trong các chất sau để khử độc thuỷ ngân?
A. Bột sắt B. Bột lưu huỳnh C. Nước D. Bột than
Câu 15: Chất nào sau đây không bị thuỷ phân trong môi trường axit?
A. Xenlulozơ B. Saccarozơ C. Fructozơ D. Tinh bột
Câu 16: Anilin có công thức hoá học là
A. C2H5NH2 B. CH3NH2 C. (CH3)2NH D. C6H5NH2
Câu 17: Khử m gam Fe3O4 bằng khí H2 ở nhiệt độ cao, thu được hỗn hợp X gồm Fe và FeO. Hỗn hợp X tác dụng vừa hết với 1,5 lít dung dịch H2SO4 0,2M (loãng). Giá trị của m là
A. 17,4 B. 23,2 C. 69,6 D. 46,4
Câu 18: Thuốc thử dùng để phân biệt bốn chất rắn: Al, Al2O3, K2O, Fe2O3 là
A. HCl B. NaOH C. H2O D. NaCl
Câu 19: Dung dịch hỗn hợp X gồm: AgNO3, Cu(NO3)2, Fe(NO3)3 (với điện cực trơ). Các kim loại lần lượt xuất hiện tại catot khi điện phân dung dịch X là:
A. Fe – Ag – Cu B. Ag – Cu – Fe
C. Fe – Cu – Ag D. Cu – Ag – Fe
Câu 20: Polime nào sau đây được tổng hợp bằng phản ứng trùng hợp?
A. Protein B. Poli(etylen terephtalat)
C. Nilon -6,6 D. Poli(vinyl clorua)
Câu 21: Kim loại nào sau đây vừa phản ứng dung dịch HCl, vừa phản ứng với dung dịch FeCl2?
A. Zn. B. Ag C. Fe D. Cu
Câu 22: Cho dãy các chất: anilin, glyxin, alanin, axit glutamic. Số chất không có tính chất lưỡng tính là
A. 2 B. 1 C. 3 D. 4
Câu 23: Khí thải công nghiệp và của các động cơ ô tô, xe máy…là nguyên nhân chủ yếu gây ra mưa axit. Thành phần hóa học chủ yếu trong các khí thải trực tiếp gây ra mưa axit là:
A. CO, NO B. CO, CO2 C. CO2, N2 D. SO2, NO2
Câu 24: Sobitol là một chất kích thích tiêu hoá, dùng tốt cho trẻ biếng ăn, rối loạn tiêu hoá do uống kháng sinh. Sobitol được điều chế bằng cách hidro hóa glucozơ. Biết hiệu suất phản ứng đạt 85%, trong quá trình điều chế có thêm 3% sobitol bị thất thoát. Khối lượng glucozơ để điều chế 50 kg sobitol thành phẩm là
A. 59,97 B. 97,95 C. 112,95 D. 49,97
Câu 25: Đốt cháy hoàn toàn 2 mol chất béo, thu được lượng CO2 và H2O hơn kém nhau 10 mol. Mặt khác, a mol chất béo trên tác dụng tối đa 450 ml dung dịch Br2 1M. Giá trị của a là
A. 0,36 B. 0,60 C. 0,40 D. 0,15
Câu 26: Cho các este: etyl fomat (1), vinyl axetat (2), triolein (3), metyl acrylat (4), phenyl axetat (5), tristearin (6). Dãy gồm các este đều phản ứng được với dung dịch NaOH (đun nóng) sinh ra ancol là:
A. (1), (3), (4), (6) B. (1), (2), (3), (4)
C. (1), (3), (4), (5) D. (2), (3), (5), (6)
Câu 27: Đốt cháy hoàn toàn m gam một amin bằng một lượng không khí vừa đủ. Trong hỗn hợp sau phản ứng chỉ có 0,4 mol CO2, 0,7 mol H2O và 3,1 mol N2. Giả sử trong không khí chỉ gồm N2 và O2 với tỉ lệ thể tích N2: O2 = 4:1. Giá trị gần nhất của m là
A. 50 B. 10 C. 5 D. 90
Câu 28: Thực hiện phản ứng nhiệt nhôm hỗn hợp X gồm Al và Fe2O3 trong điều kiện không có không khí thu được 28,92 gam hỗn hợp Y, nghiền nhỏ, trộn đều và chia hỗn hợp thành 2 phần:
- Phần 1 tác dụng vừa đủ với dung dịch NaOH dư, sau phản ứng thu được 1,008 lít khí (đktc) và 3,36 gam chất rắn không tan.
- Phần 2 tác dụng vừa đủ với 608 ml dung dịch HNO3 2,5M thu được 3,808 lít NO (đktc) và dung dịch Z chứa m gam hỗn hợp muối.
Biết các phản ứng xảy ra hoàn toàn. Giá trị m gần nhất với
A. 102 B. 100 C. 99 D. 101
Câu 29: Hỗn hợp X gồm peptit Y mạch hở (CxHyN5O6) và hợp chất Z (C4H9O2N). Lấy 0,09 mol X tác dụng vừa đủ với 0,21 mol NaOH chỉ thu được sản phẩm là dung dịch gồm C2H5OH và a mol muối của glyxin, b mol muối của alanin. Nếu đốt cháy hoàn toàn 41,325 gam hỗn hợp X bằng lượng oxi vừa đủ thì thu được N2 và 96,975 gam hỗn hợp CO2 và H2O. Giá trị a:b gần nhất với
A. 0,50 B. 0,76 C. 1,30 D. 2,60
Câu 30: Trong các thí nghiệm sau đây:
(a). Cho CO2 dư vào dung dịch Ba(OH)2
(b). Cho dung dịch HCl dư vào dung dịch Ba(HCO3)2
(c). Cho dung dịch NaOH dư vào dung dịch AlCl3
(d). Cho dung dịch HCl dư vào dung dịch Na[Al(OH)4] hoặc NaAlO2
(e). Cho CO2 dư vào dung dịch Na[Al(OH)4] hoặc NaAlO2
(f). Cho dung dịch Ca(OH)2 dư vào dung dịch Ca(HCO3)2
Số thí nghiệm cho kết tủa sau khi kết thúc phản ứng là
A. 3 B. 2 C. 4 D. 5
Câu 31: Khi nhỏ từ từ đến dư dung dịch HCl vào dung dịch hỗn hợp gồm x mol Ba(OH)2 và y mol Ba[Al(OH)4]2 [hoặc Ba(AlO2)2], kết quả thí nghiệm được biểu diễn trên đồ thị sau:
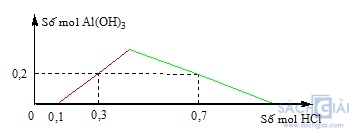
Giá trị của x và y lần lượt là:
A. 0,10 và 0,30 B. 0,05 và 0,15 C. 0,10 và 0,15 D. 0,05 và 0,30
Câu 32: Đun nóng 0,1 mol este đơn chức X với 125 ml dung dịch NaOH 1M. Sau khi phản ứng hoàn toàn, cô cạn dung dịch thu được ancol etylic và 9,2 gam chất rắn khan. Công thức cấu tạo của X là
A. C2H5COOC2H5 B. CH3COOC2H5
C. C2H5COOCH3 D. C2H3COOC2H5
Câu 33: Hỗn hợp X gồm: 0,3 mol Mg, 0,2 mol Al, 0,24 mol Zn. Cho X tác dụng với dung dịch HNO3 (loãng, dư). Sau phản ứng hoàn toàn, thấy khối lượng dung dịch tăng 28,20 gam so với dung dịch HNO3 ban đầu. Số mol HNO3 đã tham gia phản ứng gần nhất với giá trị nào?
A. 1,84 B. 1,96 C. 2,34 D. 2,16
Câu 34: Cho 7,76 gam hỗn hợp X gồm Fe và Cu (tỉ lệ số mol Fe:Cu = 7:6) tác dụng với dung dịch chứa 0,4 mol HNO3 thu được dung dịch Y và khí NO (sản phẩm khử duy nhất của N+5). Tiến hành điện phân dung dịch Y với điện cực trơ, cường độ dòng điện không đổi I = 9,65A trong thời gian t giây, thấy khối lượng catôt tăng 4,96 gam (kim loại sinh ra bám hết vào catôt). Giá trị của t là
A. 2400 B. 2000 C. 2337 D. 2602
Câu 35: Cho 27,6 gam hợp chất thơm X có công thức C7H6O3 tác dụng với 800 ml dung dịch NaOH 1M thu được dung dịch Y. Để trung hòa toàn bộ Y cần 100 ml dung dịch H2SO4 1M thu được dung dịch Z. Khối lượng chất rắn khan thu được khi cô cạn dung dịch Z là
A. 56,8 gam B. 52,4 gam C. 44,4 gam D. 58,6 gam
Câu 36: Hỗn hợp X gồm chất Y (C3H10N2O4) và chất Z (C7H13N3O4), trong đó Y là muối của axit đa chức, Z là tripeptit mạch hở. Cho 27,2 gam X tác dụng với dung dịch NaOH dư, đun nóng, thu được 0,1 mol hỗn hợp hai khí. Mặt khác 27,2 gam X tác dụng với dung dịch HCl dư, thu được m gam chất hữu cơ. Giá trị của m là
A. 44,525 B. 39,350 C. 34,850 D. 42,725
Câu 37: Cho hỗn hợp gồm a mol Mg và b mol Fe vào dung dịch chứa c mol AgNO3, khi các phản ứng xảy ra hoàn toàn thu được dung dịch X (gồm hai muối) và chất rắn Y (gồm hai kim loại). Mối quan hệ giữa a, b, c là:
A. c ≤ 2( a+b) B. 2a < c ≤ 2( a+b)
C. 2a < c < 2( a+b) D. 2(a – b) < c < 2( a+b)
Câu 38: Cho 200 ml dung dịch amino axit X nồng độ 0,4M tác dụng vừa đủ với 80 ml dung dịch NaOH 1M, thu được dung dịch chứa 10 gam muối. Khối lượng mol phân tử của X là
A. 75 B. 125 C. 103 D. 89
Câu 39: Phát biểu nào sau đây đúng?
A. Tất cả các amino axit đều làm giấy quì tím hoá đỏ
B. Chất béo là đieste của glixerol với axit béo
C. Thuỷ phân hoàn toàn tinh bột (H+, t0) thu được saccarozơ
D. Phản ứng thuỷ phân este trong môi trường kiềm là phản ứng không thuận nghịch
Câu 40: Thực hiện một số thí nghiệm với 4 chất hữu cơ thu được kết quả như sau:
| Chất Thuốc thử | X | Y | Z | T |
| Dung dịch HCl | Có phản ứng xảy ra | Có phản ứng xảy ra | Có phản ứng xảy ra | Có phản ứng xảy ra |
| Dung dịch KOH | Không xảy ra phản ứng | Không xảy ra phản ứng | Có phản ứng xảy ra | Có phản ứng xảy ra |
| Dung dịch B2 | Nước brom không bị nhạt màu | Nước brom bị nhạt màu và xuất hiện kết tủa trắng | Nước brom không bị nhạt màu | Nước brom bị nhạt màu không xuất hiện kết tủa trắng |
Các chất X, Y, Z, T lần lượt là:
A. metylamin, anilin, alanin, triolein
B. metylamin, anilin, xenlulozơ, triolein
C. etylamin, anilin, glyxin, tripanmitin
D. etylamin, anilin, alanin, tripanmitin
A. metylamin, anilin, alanin, triolein
B. metylamin, anilin, xenlulozơ, triolein
C. etylamin, anilin, glyxin, tripanmitin
D. etylamin, anilin, alanin, tripanmitin
Đáp án đề thi thử THPT Quốc gia năm 2017 môn Hóa học
| Câu | Đáp án | Câu | Đáp án | Câu | Đáp án | Câu | Đáp án |
| 1 | A | 11 | D | 21 | A | 31 | B |
| 2 | C | 12 | C | 22 | B | 32 | B |
| 3 | C | 13 | C | 23 | D | 33 | D |
| 4 | A | 14 | B | 24 | A | 34 | B |
| 5 | A | 15 | C | 25 | D | 35 | D |
| 6 | A | 16 | D | 26 | A | 36 | D |
| 7 | D | 17 | B | 27 | B | 37 | C |
| 8 | A | 18 | C | 28 | B | 38 | C |
| 9 | A | 19 | B | 29 | C | 39 | D |
| 10 | C | 20 | D | 30 | B | 40 | A |