Hướng dẫn giải bài tập SGK Hoá học lớp 10 bài 19: Luyện tập: Phản ứng oxi hóa - khử
- Thứ hai - 05/03/2018 08:33
- In ra
- Đóng cửa sổ này
Bài 1: Loại phản ứng nào sau đây luôn luôn không là loại phản ứng oxi hóa - khử?
A. Phản ứng hóa hợp.
B. Phản ứng phân hủy.
C. Phản ứng thế trong hóa vô cơ
D. Phản ứng trao đổi.
Giải:
D đúng.
Bài 2: Loại phản ứng nào sau đây luôn luôn là phản ứng oxi hóa - khử?
A. Phản ứng hóa hợp.
B. Phản ứng phân hủy.
C. Phản ứng thế trong hóa vô cơ.
D. Phản ứng trao đổi.
Giải:
C đúng.
Bài 3: Cho phản ứng: M2Ox + HNO3 → M(NO3)2 + ...
Khi x có giá trị là bao nhiêu thì phản ứng trên không thuộc loại phản ứng oxi hóa - khử?
A. x = 1.
B. x = 2.
C. x= 1 hoặc x = 2.
D. x = 3.
Chọn đáp án đúng.
Giải:
D đúng.
Bài 4: Câu nào đúng, câu nào sai trong các câu sau đây:
A. Sự oxi hóa một nguyên tố là lấy bớt electron của nguyên tố đó, là làm cho số oxi hóa của nguyên tố đó tăng lên.
B. Chất oxi hóa là chất thu electron, là chất chứa nguyên tố mà số oxi hóa của nó tăng sau phản ứng.
C. Sự khử một nguyên tố là sự thu thêm electron cho nguyên tố đó, làm cho số oxi hóa của nguyên tố đó giảm xuống.
D. Chất khử là chất thu electron, là chất chứa nguyên tố mà số oxi hóa của nó giảm sau phản ứng.
E. Tất cả đều sai
Giải:
Câu sai: B, D.
Câu đúng: A, C.
Bài 5: Hãy xác định số oxi hóa của các nguyên tố:
- Nitơ trong NO, NO2, N2O5, HNO3, HNO2, NH3, NH4Cl.
- Clo trong HCl, HClO, HClO2, HClO3, HClO4, CaOCl2.
- Mangan trong MnO2, KMnO4, K2MnO4, MnSO4.
- Crom trong K2Cr2O7, Cr2(SO4)3, Cr2O3.
- Lưu huỳnh trong H2S, SO2, H2SO3, H2SO4, FeS, FeS2.
Giải:
- Đặt x là oxi hóa của nguyên tố nitơ trong các hợp chất trên, ta có:
Trong NO: x + (-2) = 0 → x = +2.
Trong NO2: x + 2(-2) = 0 → x = +4.
Trong N2O5: 2x + 5(-2) = 0 → x = +5.
Trong HNO3: (+1) +x + 3(-3) = 0 → x = +5.
Trong HNO2: (+1) + x +2(-2) = 0 → x = +3.
Trong NH3: x + 3(+1) = 0 → x = -3.
Trong NH3Cl: x + 4(+1) +(-1) = 0 → x = -3.
- Cũng giải tương tự như trên ta có:
Bài 6: Cho biết đã xảy ra sự oxi hóa và sự khử những chất nào trong những phản ứng thế sau:
a) Cu + 2AgNO3 → Cu(NO3)2 + 2Ag.
b) Fe + CuSO4 → FeSO4 + Cu.
c) 2Na + 2H2O → 2NaOH + H2.
Giải:
Sự oxi hóa và sự khử những chất trong phản ứng thế sau:
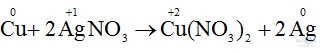
- Sự nhường electron của Cu được gọi là sự oxi hóa nguyên tử đồng.
- Sự nhận electron của ion bạc được gọi là sự khử ion bạc.
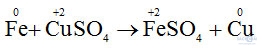
- Sự nhường electron của sắt được gọi là sự oxi hóa nguyên tử sắt.
- Sự nhận electron của ion đồng được gọi là sự khử ion đồng.
![]()
- Sự nhường electron của natri được gọi là sự oxi hóa nguyên tử natri.
- Sự nhận electron của ion hidro gọi là sự khử ion hiđro.
Bài 7: Dựa vào sự thay đổi số oxi hóa, tìm chất oxi hóa và chất khử trong những phản ứng sau:
a) 2H2 + O2 → 2H2O.
b) 2KNO3 → 2KNO2 + O2.
c) NH4NO2 → N2 + 2H2O.
d) Fe2O3 + 2Al → 2Fe + Al2O3.
Giải:
Chất khử và chất oxi hóa trong các phản ứng sau là:

Bài 8: Dựa vào sự thay đổi số oxi hóa, hãy cho biết vai trò các chất tham gia trong các phản ứng oxi hóa – khử sau:
a) Cl2 + 2HBr → 2HCl + Br2.
b) Cu + 2H2SO4 → CuSO4 + SO2 + 2H2O.
c) 2HNO3 + 3H2S → 3S + 2NO + 4H2O.
d) 2FeCl2 +Cl2 → 2FeCl3.
Giải:
Vai trò các chất trong các phản ứng oxi hóa – khử sau là:
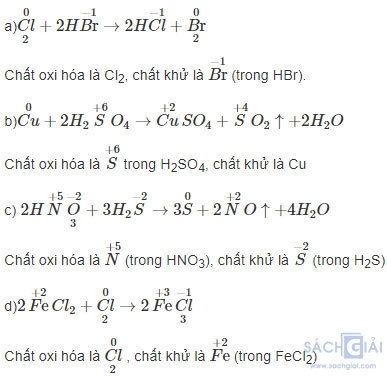
Bài 9: Cân bằng các phương trình phản ứng oxi hóa – khử sau đây bằng phương pháp thăng bằng electron và cho biết chất khử, chất oxi hóa ở mỗi phản ứng:
a) Al + Fe3O4 → Al2O3 + Fe
b) FeSO4 + KMnO4 + H2SO4 → Fe2(SO4)3 + MnSO4 + K2SO4 + H2O
c) FeS2 + O2 → Fe2O3 + SO3
d) KClO3 → KCl + O2
e) Cl2 + KOH → KCl + KClO3 + H2O
Giải:
Cân bằng các phương trình phản ứng oxi hóa – khử:

Bài 10: Có thể điều chế MgCl2 bằng:
- Phản ứng hóa hợp.
- Phản ứng thế.
- Phản ứng trao đổi.
Giải:
Điều chế MgCl2 bằng:
- Phản ứng hóa hợp: Mg + Cl2 → MgCl2
- Phản ứng thế: Mg + CuCl2 → MgCl2 + Cu
- Phản ứng trao đổi: Mg(OH)2 + 2HCl → MgCl2 + 2H2O
Bài 11: Cho những chất sau: CuO, dung dịch HCl, H2, MnO2.
a) Chọn từng cặp trong những chất đã cho để xảy ra phản ứng oxi hóa – khử và viết phương trình phản ứng.
b) Cho biết chất oxi hóa, chất khử, sự oxi hóa và sự khử trong những phản ứng hóa học nói trên.
Giải:
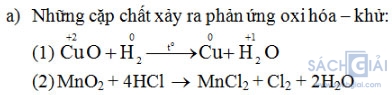
b) Trong phản ứng (1):
- Nguyên tử hiđro nhường electron là chất khử, sự nhường electron của H2 được gọi là sự oxi hóa nguyên tử hiđro.
- Ion Cu nhận electron, là chất oxi hóa. Sự nhận electron của ion đồng được gọi là sự khử ion đồng.
Trong phản ứng (2):
- Ion Clo nhường electron là chất khử. Sự nhường electron của Cl- được gọi là sự oxi hóa ion Clo.
- Ion Mn nhận electron là chất oxi hóa. Sự nhận electron của ion Mn được gọi là sự khử ion Mn.
Bài 12: Hòa tan 1,39g muối FeSO4.7H2O trong dung dịch H2SO4 loãng. Cho dung dịch này tác dụng với dung dịch KMnO4 0,1M. Tính thể tích dung dịch KMnO4 tham gia phản ứng.
Giải:
Phương trình hóa học của phản ứng:
10FeSO4 + 2KMnO4 + 8H2SO4 → 5Fe2(SO4)3 + K2SO4 + 2MnSO4 + 8H2O.
nFeSO4.7H2O = 1,39 / 278 = 0,005 mol = nFeSO4.
nKMnO4 = 2nFeSO4 = 0,01 mol.
Vdd KMnO4 = 0,001 / 0,1 = 0,01 (lit).
